



Redox Reactions

Involve exchange of electrons. 1 substance must be oxidised and the other reduced.
One substance Second substance
Loses electrons - oxidised Gains electrons - reduced


Oxidising & Reducing Agents
Usually a metal/ negative Ion that is itself oxidised.
Donates electrons to a 2nd substance acting as an agent REDUCING the 2nd substance.
Usually a non metal/positive Ion that is itself reduced
Gains electron from a 2nd substance acting as an agent OXIDISING the 2nd substance.




Oxidising/Reducing Agent Half Equations

1. Metal atom (lose electrons) 2. Negative Ion (lose electrons)
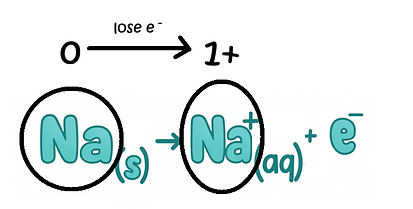

reducing
agent
reducing
agent

1.Non metal atom (gain electrons) 2. Positive Ion (gain electrons)


oxidising
agent
oxidising
agent
Hydrogen - Oxidising & Reducing Agent
1.. Turn alcohols into aldehydes/ketone then carboxylic acids in the lab.
2. Kill fungi and bacteria & inactivate viruses.
2. Break down coloured compounds in hair/clothes (hydrogen peroxide)
Uses of Oxidising Agents
1.. Turn alcohols into aldehydes/ketone then carboxylic acids in the lab.
2. Kill fungi and bacteria & inactivate viruses.
2. Break down coloured compounds in hair/clothes (hydrogen peroxide)
Best Reducing & Oxidising Agents
The electrochemical series represents a series of reduction reactions found in the Data Booklet.
Strongest reducing agents
Alkali metals (Group 1) as they have low electronegativities and lose electrons very easily.
Halogens (group 7) which are highly electronegative and gain electrons easily.
Easier Redox Equations


Harder Redox Equations
1. Balance the atom present on both sides.
2. If oxygen atoms are not balanced, add t water molecules to the side with the lower oxygen.
3. Add twice the number of hydrogen ions as water molecules to the OTHER side
(side with original higher oxygen atoms).
4. Add electrons to balance the charge on both side.