


Trends in Periodic Table


Measures half the distance between the nuclei of 2
covalently bonded atoms of an element.


The energy required to remove 1 mole of electrons
from 1 mole of gaseous atoms.


The attraction an atom in a bond has for the
electrons in a bond.



Periodic Table
Trends across a period/row
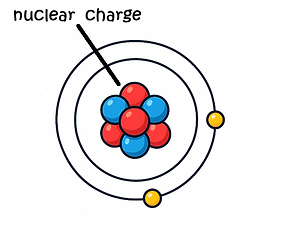
Increased nuclear charge (protons)
This causes outer electrons to be pulled closer.
Trends down a group
Increased screening effect ( 1 mark)
More shells so attraction of nucleus/protons for outer electrons decrease
(1 mark)

Noble Gases
1. No Covalent radius
2.No Electronegativity
They have full shells hence don't form
covalent bonds.
3. Highest ionisation energy values

Ionisation energy



Always an endothermic reaction =
1st Ionisation Energy
2nd Ionisation Energy
3rd Ionisation energy

Ionisation energy Calculation

To work out 2nd ionisation energy you
need to
total 1st + 2nd.
578 + 1817 = 2395
To work out 3rd ionisation energy you
need to
total 1st + 2nd + 3rd.
578 + 1817 + 2745 = 5140
Ionisation energy Graph Rows
Peaks = Noble Gases Lowest point = Alkali metals

Ionisation energy Graphs Groups
Group 2 Ionisation energy
Group 1 Ionisation energy


e.g 2, 8, 1
e.g 2, 8, 2

e.g 2, 8, 3
Higher Ionisation Energy
Removing electrons from an inner/full shell results in significantly higher ionisation energy values.


The 2nd/3rd ionisation energy is significantly higher than the first ionisation energy.
This tell us the element in this example in group 1.
Removing a second outer electron requires it being removed from an inner shell
Group 3 Ionisation Energy
Electron Affinity
Ionisation Energy & Electron Affinity
Watch out for problem solving questions about electron affinity.
It is the REVERSE of ionisation energy

Ionisation Energy
Remove 1 mole of electrons
Electron affinity
Gain 1 mole of electrons
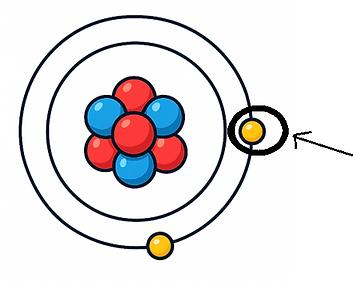


Atomic vs Ionic Radius

Metal atoms larger than ions Non metal atoms smaller than ions
Metal Ionic Radius
When metal atoms become metal ions they LOSE electrons = lose an energy shell.
Larger Metal atoms Smaller Metal ions
More shells 2, 8, 1 Less Shells 2, 8


